Acids and Bases
• The word acid comes from the Latin word acere which means sour.
• Curd, lemon juice, orange juice, amla, and vinegar taste sour because they contain acids(natural acids).
• The chemical nature of such substances is acidic.
• Baking soda, does not taste sour.
• That means, it has no acids in it.
• It is bitter in taste.
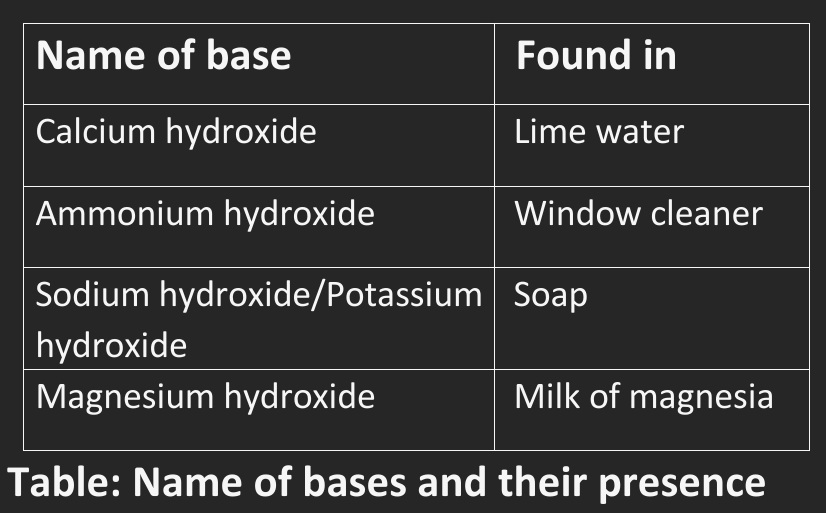
• The substances like these which are bitter in taste and feel soapy on touching are known as bases.
• And the nature is called basic.
Table: Various substances and their taste

Table: Name of naturally occurring acids and their presence
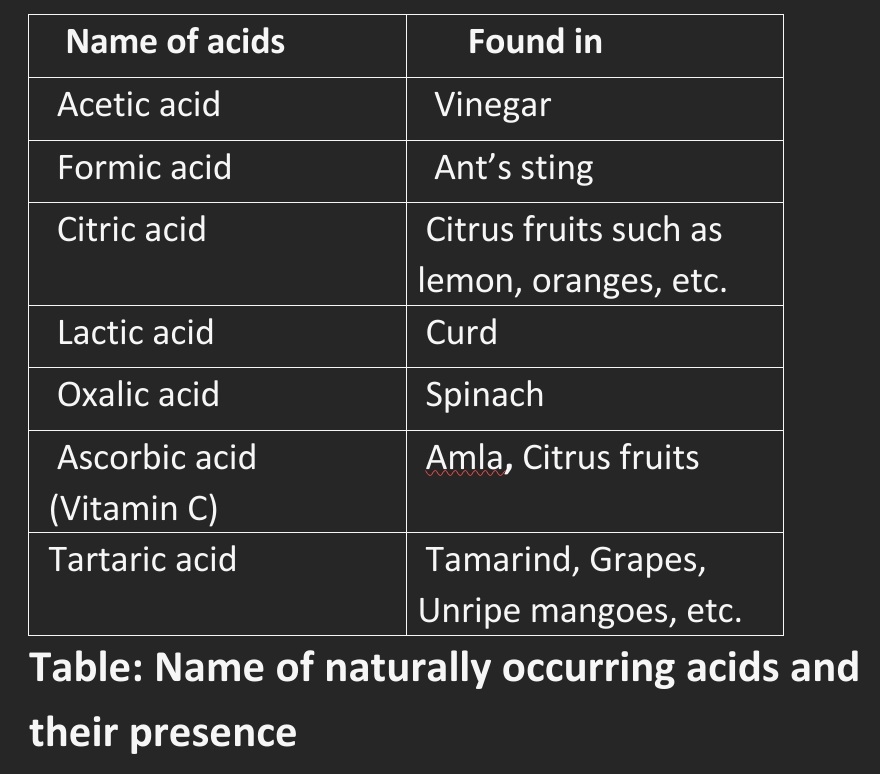
Table: Name of bases and their presence
Indicators
• The substances are used to test whether a substance is acidic or basic are known as indicators.
• They change their colour when added to a solution of acidic or a basic substance.
• Examples: Turmeric, litmus, China rose petals (Gudhal), etc., are the naturally occurring indicators.
Natural Indicators Around Us:
Litmus: A natural dye
• The most common natural indicator is litmus, extracted from lichens.
• The colour of litmus is mauve (purple) in distilled water.
• When added to an acidic solution, it turns red.
• When added to a basic solution, it turns blue.
• Generally, available in the form of a solution, or in the form of strips of paper, known as litmus paper in red and blue colour.
• The solutions which do not change the colour(either red or blue litmus), known as neutral solutions.
• These substances are neither acidic nor basic.
Lime water is the common name for a diluted solution of calcium hydroxide [Ca(OH)₂]
China Rose Indicator
Take Some china rose (Gudhal) petals in a beaker and add some warm water in it. After some time water becomes coloured. That coloured water is China Rose indicator.
• China rose indicator turns acidic solutions to dark pink (magenta).
• And turns basic solutions to green.
Lime water
Lime water is the common name for a diluted solution of calcium hydroxide [Ca(OH)₂]
Acid Rain
• The rain containing excess of acids is called an acid rain.
• The rain becomes acidic because carbon dioxide, sulphur dioxide and nitrogen dioxide (which are released into the air as pollutants) dissolve in rain drops to form carbonic acid, sulphuric acid and nitric acid respectively.
• It may cause damage to buildings, historical monuments, plants and animals.
Neutralisation
• When an acidic solution is mixed with a basic solution, both neutralise the effect of each other.
• After mixing in suitable amounts, both the nature(acidic and basic) are destroyed.
• The resulting solution is neither acidic nor basic.
• The neutralisation reaction can be defined as:
The reaction between an acid and a base is known as neutralisation. Salt and water are formed in this process with the evolution of heat.
Acid + Base → Salt + Water(Heat is evolved)
Example:
Hydrochloric acid (HCl) + Sodium hydroxide (NaOH) →Sodium chloride (NaCl) + Water (H2O)
Neutralisation in Everyday Life
Indigestion
• Our stomach contains hydrochloric acid which helps to digest food.
• But excessive acid in the stomach causes indigestion(sometimes painful).
• To relieve indigestion, we take an antacid such as milk of magnesia, which contains magnesium hydroxide.
• It neutralises the effect of excessive acid.
Ant bite
• When an ant bites, it injects the acidic liquid (formic acid) into the skin.
• Acid effect can be neutralised by rubbing moist baking soda (sodium hydrogencarbonate) or calamine solution(contains zinc carbonate).
Soil treatment
• Excessive use of chemical fertilisers makes the soil acidic.
• It is harmful for plants.
• Plants do not grow well when the soil is either too acidic or too basic.
• When it is too acidic, it is treated with [quick lime (calcium oxide)] or [slaked lime (calcium hydroxide)].
• If it is basic, organic matter (compost) is added to it which releases acids to neutralises the basic nature of the soil.
Factory wastes
• The wastes coming from many factories contain acids.
• If they are flow into the water, the acids will kill fish and other organisms.
• Therefore, factory wastes are neutralised by adding basic substances.

Leave a comment