SHORT NOTES
Physical changes :-
- Properties like shape, size, colour and state of a substance are called its physical properties.
- A change in which a substance undergoes a change in its physical properties is called a physical change.
- A physical change is generally reversible.
- In such a change no new substance is formed.
- Examples :-
- Melting of Ice
- Evaporation of water.
- Dissolution of sugar in water.
All these processes do not lead to the formation of new chemical substances. For example,
- Ice, water, and water vapour are chemically same. i.e., water (H2O).
- Solution of sugar in water still remains the same chemical substances, that is , sugar and water.
Chemical changes :-
Processes in which the original substances lose their nature and identity and form new chemical substances with different properties are called chemical changes.
Example :-
- Rusting of iron articles especially in the rainy season.
- Souring of milk in summer
- Burning of coke in air.
In all these processes, the original substances lose their nature and identity, and form new chemical substances.
For example :–
- Rust is chemically a different compound than the original substance, iron.
- Similarly, we cannot drink milk which has turned sour because the properties of milk have changed to form some new substances.
The process involving a chemical change is called a chemical reaction.
What is chemical ?
A chemical is any substance that has a defined composition. In other words, a chemical is always made up of the same “atoms”.
Some chemicals occur in nature, such as water. Other chemicals are manufactured, such as chlorine (used for bleaching fabrics or in swimming pools).
Chemicals are all around you: the food you eat, the clothes you wear. You, in fact, are made up of a wide variety of chemicals.
What is chemical reaction ?
- A chemical reaction refers to a change in a chemical.
- More generally, a chemical reaction can be understood as the process by which one or more substances change to produce one or more different substances.
- During a chemical reactions, atoms of one element do not change into the atoms of another element.
Examples of chemical reactions occurs in daily life:
• Souring of milk
• Formation of curd from milk
• Cooking of food
• Digestion of food in our body
• Process of respiration
• Rusting of iron
• Burning of fuels
• Burning of candle wax
• Decomposition of waste
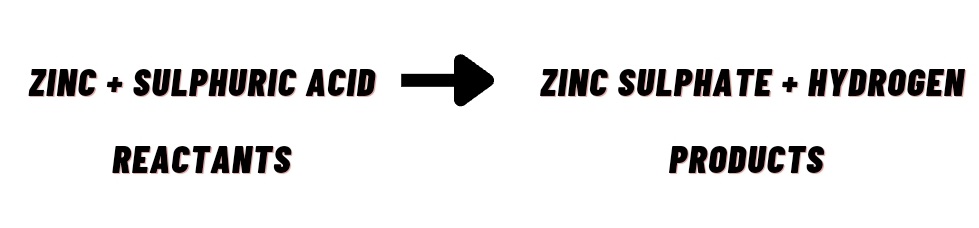
What are reactants ?
The substance which takes part in chemical reaction are called reactants.
What are products ?
The new substance produced as a result of chemical reactions.
Why the magnesium ribbon is cleaned by rubbing with sand paper before burning in air ?
- This is done to remove the protective layer of magnesium oxide from the surface of magnesium ribbon before burning in air.
Characteristics of chemical reactions :
- When a chemical reaction takes place, some changes are observed.
- The easily observable changes that take place in a chemical reaction are called the characteristics of the chemical reaction.
- These changes help us to check that a chemical reaction has taken place.
- Some important characteristics of chemical reactions are given below:
- Evolution of gas
- Formation of precipitate
- Change in colour
- Change in temperature
- Change in state
EVOLUTION OF GAS
Some chemical reactions are accompanied by the evolution of
gas.
For example:-
- (i) In the reaction between zinc and dilute sulphuric acid or hydrochloric acid, hydrogen gas is evolved.
- (ii) When calcium carbonate is heated, or calcium carbonate reacts with dilute hydrochloric acid, carbon dioxide is evolved.
FORMATION OF PECIPITATE
- Some chemical reactions are accompanied by the formation of a precipitate.
- A precipitate is a solid substance formed on mixing two substances.
For example :-
- (i) In the reaction between lead nitrate and potassium iodide solutions, a yellow precipitate of lead iodide appears.
- (ii) When silver nitrate solution is mixed with sodium chloride solution, a white precipitate of silver chloride is obtained.
- (iii) When barium chloride solution is mixed with sodium sulphate solution, a white precipitate of barium sulphate is obtained.
CHANGE IN COLOUR
Some chemical reactions are accompanied by change in color.
For example :-
- (i) Rust is brown in colour whereas iron is greyish black in colour.
- (ii) Lead nitrate and potassium iodide solutions are colourless, but on mixing the two, the precipitate of lead iodide formed is yellow in color.
CHANGE IN STATE
Some chemical reactions are accompanied by change in state.
For example :-
- (i) Solid wax (in the form of candle) burns to form water vapour and carbon dioxide which are gaseous.
- (ii) Petrol, which is a liquid, burns to form water vapour and carbon dioxide which are gaseous.
CHANGE IN TEMPERATURE
Some chemical reactions are accompanied by change in
temperature.
Two ways in which temperature can change
- Rise of temperature.
- Fall of temperature.
Reactions which are accompanied by rise in temperature are those in which heat is involved. Such reactions are called exothermic reactions.
Examples of Exothermic reaction :-
(i) When zinc pieces react with sulphuric acid in a beaker or a flask, it is found
to be warm. This can be seen by touching the beaker or the flask.
(ii) When water is added into quick lime (CaO), taken in a beaker, slaked lime
Ca(OH)₂ , is formed and the beaker is found to be quite hot.
Reactions which are accompanied by fall in temperature are those in which heat is absorbed. Such reactions are called endothermic reactions.
Examples of Endothermic reactions :-
When barium hydroxide, Ba(OH)₂, is added into ammonium chloride, NH4Cl, taken in a test tube, and mixed with a glass rod, then on touching the bottom of the test tube, it is found to be cooler.
This is due to the following
endothermic reaction taking place:
Barium Hydroxide +Ammonium chloride ——-> Barium chloride + Ammonium hydroxide.
Reactions involve several changes simultaneously. It is important to know that in several reactions; more than one change may be observed. i.e., they may possess more than one characteristic.
For example,
(i) When zinc reacts with dilute sulphuric acid, a gas i.e. Hydrogen is evolved and a rise in temperature takes place.
Chemical Equation :-
The short-hand method of representing a chemical reaction in terms of symbols and formulae of the different reactants and products is called a chemical equation.
There are two ways of representing a chemical reaction as follows:
- (i) In terms of words (called word equation).
- In terms of symbols and formulae (called chemical equation).
For example,
(i) Word equation can be represented as follows:
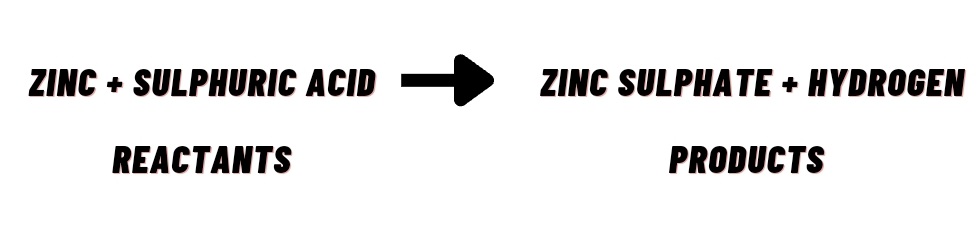
(ii) Chemical equation can be represented as follows:

Balancing of a Chemical Equation
Balancing of a chemical equation means making the number of atoms of each element equal on both sides of the equation.
Number of atoms on LHS of equation = Number of atoms on RHS of equation
Steps involved in the balancing of a chemical equation:
- Write down the equation in the word form.
- Write down the symbols and formulas of the various reactants and products.
- Make the number of atoms of each element equal on both sides of the equation.
TYPES OF CHEMICAL REACTIONS :-
Combination reaction:-
Combination reactions – Reactions in which two or more reactants combine to
form one product is called Combination Reactions.
1) 2Mg(s) + O₂(g) → 2MgO
2) 2H₂ (g) + O₂(g)→ 2H₂O (l)
3) C(s) + O₂ (g)→ CO₂(g)
4) H₂ (g) + Cl2 (g)→ 2HCl
5) 2Na + Cl → 2NaCl
6)Fe + S → FeS
7) CaO + H₂O →Ca(OH)₂
8) NH₃ + HCl → NH4Cl
9) 2CO + O₂ → 2CO₂
10) 2SO₂ + O₂ →2SO3
Decomposition reaction:-
Decomposition reactions – Reactions in which one compound decomposes into
two or more compounds or elements are known as Decomposition Reaction.
Examples :-
1) CaCO3→ CaO + CO₂
2) 2Pb(NO₃)₂ → 2PbO + 4NO₂ + O₂
3) 2FeSO₄ → Fe2O3 + SO₂ + SO3
4) 2KClO3 → 2KCl +3O₂
5) C2H2O4.H₂O → C2H2O4 + H₂O
6) 2AgCl → 2Ag + Cl2
(Photodecomposition reaction)
7) 2AgBr → 2Ag+ Br2
(photodecomposition reaction)
8) 2H₂O₂ → 2H₂O + O₂ (In presence of light)
Displacement reaction :-
Displacement reactions – The chemical reaction in which a more reactive element displaces a less reactive element from a compound is known as a displacement reaction.
Examples :-
1) CuSO₄ + Zn → ZnSO₄ + Cu
2) CuSO₄ + Fe → FeSO₄ + Cu
3) CuSO₄ + Mg → MgSO₄ + Cu
4) 2Na + 2H₂O → 2NaOH+ H₂
5) CuO + Mg → MgO + Cu
6) CuCl2 + Pb → PbCl2 + Cu
7) Fe₂O₃ + 2Al→ 2Fe + Al₂O₃
8) Fe + 2HCl → FeCl2 + H₂
9) Cl2 + 2KI → 2KCl + I2
10) Mg +2HCl → MgCl2 + H₂
Double displacement:-
Double displacement reactions – Reactions in which ions are exchanged between two reactants forming new compounds are called Double Displacement Reactions.
Examples :-
1) AgNO3 + NaCl → AgCl + NaNO3
2) BaCl2+ Na2SO₄ → BaSO₄ + 2NaCl
3) CuSO₄ + H₂S → CuS + H₂SO₄
4) AlCl₃ + 3NH4OH → Al(OH)3 + 3NH4Cl
5) BaCl2 + CuSO₄ → BaSO₄ + CuCl2
6) NaOH + HCl → NaCl + H₂O
7) Pb(NO₃)₂ + 2KI → PbI2 + 2KNO3
8) CuSO₄ + 2NaOH → Cu(OH)₂ + Na2SO₄
9) CoCl2 + Na2CO3 → CoCO3 + 2NaCl
10) Pb(C2H3O2)2 + 2HCl → PbCl2 +
2CH₃COOH
Precipitation reactions :-
Precipitation reactions – The reaction in which precipitate is formed by the mixing of the aqueous solution of two salts is called precipitation reaction.
Example : AgNO3 + NaCl → AgCl + NaNO3
Neutralization Reaction :-
Neutralization Reaction – The reaction in which an acid reacts with a base to form salt and water by an exchange of ions is called Neutralization Reaction.
Example: NaOH + HCl → NaCl + H₂O
Oxidation :-
Oxidation – Addition of oxygen or non-metallic element or removal of hydrogen or metallic element from a compound is known as oxidation.
Reduction :-
Reduction – Addition of hydrogen or metallic element or removal of oxygen or
non-metallic element from a compound is called reduction.
The compound or element which undergoes reduction is called to be reduced.
Oxidizing agent:
• The substance which gives oxygen for oxidation is called an Oxidizing agent.
• The substance which removes hydrogen is also called an Oxidizing agent.
Reducing agent:
• The substance which gives hydrogen for reduction is called a reducing agent.
• The substance which removes oxygen is also called a reducing agent.
Redox reactions :-
Redox reactions – A chemical reactions where oxidation and reduction both take
place simultaneously are also known as redox reaction.
Exothermic reactions :-
Exothermic reactions – Reactions which produce energy are called exothermic
reaction. Most of the decomposition reactions are exothermic.
Example :-
(i)Respiration is an exothermic reaction in which energy is released.
C6H12O6 (s) + 6 O2 (g) → 6 CO2 (g) + 6 H2O (l) + heat
ΔG = −2880 kJ per mol of C6H12O6
(ii) When quick lime (CaO) is added to water, it releases heat energy.
Example :-
CaO + H₂O →Ca(OH)₂ + heat
Endothermic reactions :-
Endothermic reactions – Reactions which absorb energy are called endothermic reaction. Most of the combination reactions are endothermic.
Example :-
(i) Decomposition of Calcium Carbonate.
CaCO3→ CaO + CO₂
Note : Inert gas (Ne, Ar) elements do not react with other elements.

Leave a comment